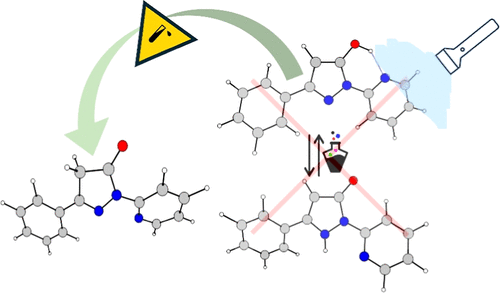
The tautomeric behavior of 1-(2-pyridinyl)-1H-pyrazol-5-ols has been investigated in solution using UV–vis and NMR spectroscopies and DFT quantum-chemical calculations. The study has been focused on the influence of the molecular structure, solvents, and acid addition protonation. The tautomerism has been considered in light of the possible long-range intramolecular proton transfer and tautomeric switching under irradiation. The results demonstrate that the possibilities for photoswitching are limited due to specific structural factors preventing population and stabilization of the end keto tautomer. At the same time, a very rare case of a middle-range OH/CH proton transfer can be achieved by acid addition.
https://doi.org/10.1021/acs.jpcb.5c08439
